Study Overview
Importantly, Dr. Nikhil Bharatbhai Patel and colleagues conducted this prospective study to examine clinical parameters and outcomes of early intravenous thrombolysis using recombinant tissue plasminogen activator (r-tPA) in stroke patients. Furthermore, the research took place at Jehangir Hospital, Pune’s oldest tertiary care center, which handles numerous stroke cases annually.
Research Objectives
- First, implement emergency protocols for immediate patient triage of all stroke patients
- Second, conduct a timeline analysis to review thrombolysis outcomes using NIHSS (National Institutes of Health Stroke Scale) and MRS (Modified Rankin Scale) scores at discharge
- Third, achieve process improvement by identifying door-to-needle and door-to-CT times with and without code stroke implementation
- Finally, perform a clinical assessment to evaluate baseline parameters in acute stroke patients
Study Design & Methodology
Patient Population
Initially, the research included adult patients (>18 years) who presented with sudden-onset stroke symptoms. Additionally, both male and female patients from Pune’s urban population participated in the study.
Sample Groups
Subsequently, researchers organized participants into two distinct groups:
- Code stroke implementation group: 52 patients
- Non-implementation group: 48 patients
Moreover, researchers calculated the sample size using established statistical methods, targeting 80% power at a 5% significance level to detect meaningful differences in NIHSS scores.
Study Timeline
Importantly, the research was divided into two distinct periods:
- Pre-code stroke era: April 2015 to July 2015 (protocol not implemented)
- Code stroke era: August 2015 to April 2017 (protocol active)
Data Collection Methods
Specifically, medical teams recorded comprehensive patient information, including:
- Demographics (age, gender)
- Medical history and risk factors
- Initial stroke severity assessment
- Critical time points: symptom onset, hospital arrival, neuroimaging, treatment administration
Data collection technique and tools:
- Furthermore, we implemented the ‘SC’ protocol in August 2015. Consequently, the study time period was divided into the ‘pre-SC era’ (April 2015 to July 2015), in which the protocol was not implemented, and the ‘SC era’ (August 2015 to April 2017), in which stroke code was implemented. Regardless of SC activation, patients who arrived at the ED within 4.5 hours of stroke onset were identified. Subsequently, demographic data (age and gender), medical history, and vascular risk factors, including hypertension, diabetes mellitus, atrial fibrillation, ischemic heart disease, valvular heart disease, and smoking, were recorded. Additionally, initial stroke severity was assessed by the NIHSS.
- Next, acute ischemic stroke patients who received IV r-tPA were extracted from the registry and specially encoded in another database. Specifically, the important time points, which include stroke symptoms onset (onset time), presentation to the ED (door time), neuroimaging (CT time), and bolus IV r-tPA administration (needle time), were all specifically recorded by the nursing practitioner. Furthermore, stroke characteristics and outcomes were recorded. Subsequently, follow-up brain imaging by CT or magnetic resonance imaging was routinely performed 24 to 36 hours after IV r-tPA administration to ascertain whether ICH had occurred. Notably, symptomatic ICH was defined as a neurological deterioration (NIHSS > 2 points) occurring within 36 hours and with no radiological findings that might have been responsible for this deterioration other than hemorrhage. Finally, outcomes were assessed by the Modified Rankin Scale (MRS) score at discharge.
- However, a good outcome occurred when the MRS score was <3. In addition, we also analyzed for in-hospital mortality. Specifically, we used an informed consent form, a consent form for IV r-tPA administration, an NIHSS evaluation sheet, a checklist for inclusion and exclusion criteria for IV r-tPA thrombolysis, and a patient information sheet to collect the data
Data analysis:
Initially, the data on categorical variables is shown as n (% of cases), and the data on continuous variables is presented as Mean and Standard deviation (SD) if it is normally distributed. Otherwise, the median (min – max) is used across two study groups. Furthermore, the inter-group comparison of categorical variables is done using the Chi-square test / Fisher’s exact probability test.- Additionally, the statistical significance of inter-group difference of mean of continuous variables is tested using unpaired OR independent sample ‘t’ test for normally distributed variables. Conversely, the Mann-Whitney U test is used for comparing non-normally distributed variables across two study groups. Before analysis, the underlying normality assumption was tested before subjecting the study variables to the t-test. Subsequently, the entire data is entered and cleaned in MS Excel before its statistical analysis.
- Moreover, all the results are shown in tabular as well as graphical format to visualize the statistically significant difference more clearly. Importantly, the p-values less than 0.05 are considered to be statistically significant. Additionally, all the hypotheses are formulated using two-tailed alternatives against each null hypothesis (hypothesis of no difference). Finally, the entire data is statistically analyzed using Statistical Package for Social Sciences (SPSS ver 16.0, Inc., IBM Corporation) for MS Windows.
Key Findings
Patient Demographics
Initially, the study revealed several important patterns:
- First, no significant age difference existed between groups
- Additionally, stroke incidence increased with age in both sexes
- Furthermore, males comprised the majority of patients in both groups
Risk Factor Analysis
Subsequently, common comorbidities among patients included:
- Most notably, hypertension: 60% of patients
- Additionally, diabetes: 39% of patients
- Furthermore, previous ischemic heart disease: 21% of cases
- Moreover, smoking history: 23% of patients
- Finally, atrial fibrillation: 16% of patients
Treatment Time Improvements
Door-to-Needle Time (DTNT)
Significantly, the code stroke implementation dramatically reduced treatment delays:
- In contrast, the non-implementation group: Mean 37.5 minutes (median 28.0 minutes)
- However, implementation group: Mean 21.2 minutes (median 17.5 minutes)
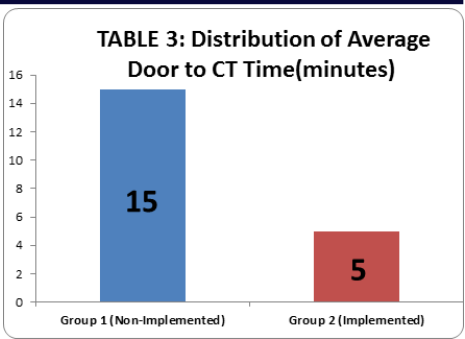
Door-to-CT Time (DTCT)
Similarly, imaging times also improved significantly:
- Specifically, non-implementation group: Mean 18.4 minutes (median 15.0 minutes)
- In comparison, implementation group: Mean 5.5 minutes (median 5.0 minutes)
Clinical Outcomes
Patient Recovery
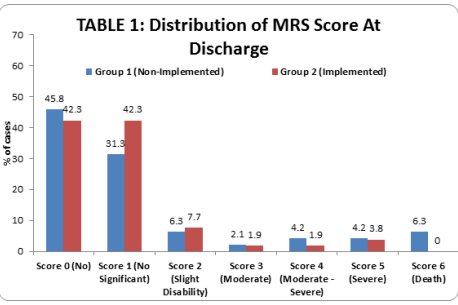
Notably, favorable discharge outcomes (MRS score 0-1) increased substantially:
- Previously, the non-implementation group: 77.08% of patients
- Subsequently, the implementation group: 84.61% of patients
Safety Metrics
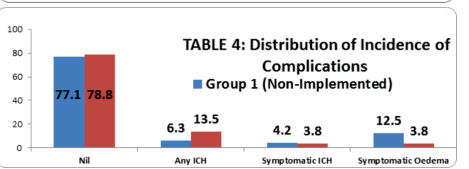
- Hospital mortality: Zero deaths in the implementation group
- Symptomatic intracranial hemorrhage: Decreased from 4.2% to 3.8%
- Mean NIHSS at discharge: 2.0 points in the implementation group
Quality Targets Achievement
The implementation group successfully met critical benchmarks:
Door-to-needle time <60 minutes: Achieved in 100% of patients
Door-to-CT time <25 minutes: Achieved in 100% of patients
Clinical Implications
Stroke Location Patterns
Anatomical distribution showed:
- Middle cerebral circulation: 76% (most common)
- Posterior circulation: 22%
- Anterior circulation: 2%
Imaging Results
CT scan findings revealed:
- Normal findings: 39% of thrombolyzed patients
- Early infarction signs: 31% of patients
- Hyperdense MCA sign: 21% of patients
Statistical Analysis Framework
Researchers employed rigorous analytical methods using SPSS version 16.0. They analyzed categorical variables with chi-square tests and continuous variables with appropriate parametric or non-parametric tests based on data distribution. Statistical significance was set at p<0.05.
Conclusion
Implementation of AHA/ASA-recommended strategies effectively reduced door-to-needle times in this private tertiary care setting. The protocol improvements led to:
- Enhanced treatment speed: Significant reductions in both door-to-needle and door-to-CT times
- Better patient outcomes: Improved NIHSS and MRS scores at discharge
- Increased safety: Reduced mortality and hemorrhage complications
- Quality achievement: Met all targeted time benchmarks
Furthermore, these results demonstrate that structured stroke protocols can substantially improve emergency care delivery and patient outcomes in acute ischemic stroke treatment. The findings support broader implementation of similar emergency response systems in tertiary care hospitals.
References
- Jauch EC, Saver JL, Adams HP Jr, et al ; American Heart Association Stroke Council; Council on Cardiovascular Nursing; Council on Peripheral Vascular Disease; Council on Clinical Cardiology.Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke.2013;44(3):870-947.
- Tissue plasminogen activator for acute ischemic stroke: the National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995;333(24):1581-1587.
- Wardlaw JM, Murray V, Berge E, et al. Recombinant tissue plasminogen activator for acute ischaemic stroke: an updated systematic review and meta-analysis. Lancet.2012;379(9834):2364-2372.
- Lees KR, Bluhmki E, von Kummer R, et al; ECASS, ATLANTIS, NINDS and EPITHETrt-PAStudy Group. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet. 2010;375(9727):1695-1703.
- Marler JR, Tilley BC, Lu M, et al. Early stroke treatment associated with better outcome: the NINDS rt-PAstroke study. Neurology.2000;55(11):1649-1655.
- Lansberg MG, Schrooten M, Bluhmki E, Thijs VN,Saver JL. Treatment time-specific number needed to treat estimates for tissue plasminogen activator therapy in acute stroke based on shifts over the entire range of the modified Rankin Scale. Stroke.2009;40(6):2079-2084.
- Saver JL, Fonarow GC, Smith EE, et al. Time to treatment with intravenous tissue plasminogen activator and outcome from acute ischemic stroke. JAMA. 2013;309(23):2480-2488.
- Fonarow GC, Smith EE, Saver JL, et al.Timeliness of tissue-type plasminogen activator therapy in acute ischemic stroke: patient characteristics, hospital factors, and outcomes associated with door-to-needle times within 60 minutes. Circulation. 2011;123(7):750-
Export Full Research Report As a PDF File